DNA Logic Gates as Components of Molecular Robots

A molecular robot is a next-generation biochemical machine that imitates the actions of microorganisms. It is made of biomaterials such as DNA, proteins, and lipids. Three prerequisites have been proposed for the construction of such a robot: sensors, intelligence, and actuators. This Mini review focuses on recent research on synthetic ion channels and DNA computing technologies, which are viewed as potential candidate components of molecular robots. Synthetic ion channels, which are embedded in artificial cell membranes (lipid bilayers), since ambient ions or chemicals and import them. These artificial sensors are useful components for molecular robots with bodies consisting of a lipid bilayer because they enable the interface between the inside and outside the molecular robot to function as gates. After the signal molecules arrive inside the molecular robot, they can operate DNA logic gates, which perform computations. These functions will be integrated into the intelligence and sensor sections of molecular robots. Soon, these molecular machines will be able to be assembled to operate as a mass micro robot and play an active role in environmental monitoring and in vivo diagnosis or therapy.
1. Molecular Robots and the Lipid Bilayer Platform
Molecular robots have recently emerged based on biomolecules and biochemical processes. Approximately 30 years ago, a self-constructing machine, a so-called “assembler,” was originally proposed by Drexler. Based on the idea of an assembler, molecular machines that operate autonomously have been developed using DNA or RNA. For example, DNA walkers move autonomously, on the basis of energy supplied from the hybridization of fuel oligonucleotides, from one binding site to another on a DNA-modified surface. Rothemund has also proposed a method by which DNA molecules can be folded into any desired two-dimensional shape to make “DNA origami.” In the field of synthetic chemistry, nano sized machines such as motors and ratchets have been developed based on organic or supramolecular chemistry. This was viewed as ground-breaking technology and the pioneers have since been honoured with the Nobel Prize in Chemistry 2016. Inspired by the idea of a molecular assembler, molecular robotics, which involves construction with a much higher dimension of assembly, was proposed in 2014. Molecular robots are
composed of sensors, calculators, and actuators that are all implemented in liposomes or hydrogels. White blood cells, the most imageable example, senses the chemical signals secreted from a target bacterium, calculates the direction and length between the target and itself, and moves toward the signal source by chemotaxis. These accomplished functions are integrated in a micron-sized body surrounded with a bilayer lipid membrane (BLM). Sato et al. reported the development of a sophisticated molecular robot prototype in 2017. Their developed amoeba-type robot has light-induced DNA clutches for sensors and kinesin-microtubule proteins as actuators, all integrated in a cell-sized liposome. Light irradiation acts as a trigger for the release of the signal molecules and disengagement of the DNA clutches to change the shape of the liposome. The fabrication process used for existing mechanical robots is viewed as a blueprint for the manufacture of these molecular robots. In the case of humanoid robots, the arms and legs are manufactured individually and then assembled. Similarly, individual fabrication would be a straightforward process in the manufacturing of molecular robots.
2. Synthetic Ion Channels and Transport Control at the Lipid Bilayer Membrane
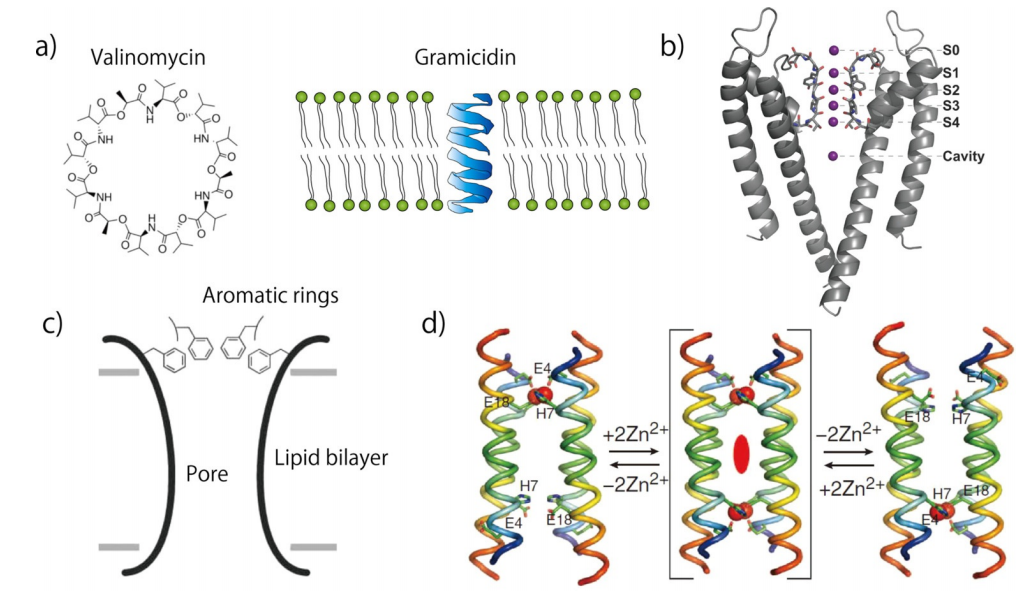
Receptor proteins sense specific molecules in the cell membrane. When the ligand molecule binds to a receptor, the receptor senses the binding and produces a signal that is transmitted downstream to control ion transport through ion channels, as it cascades. Furthermore, pore-forming proteins play a key role in the transportation of molecules with the gradient of membrane potential or the substrate concentration. Artificial ion channels or pores have been created using synthetic chemistry to mimic the structure or function of natural proteins. In the last three decades, synthetic chemists have proposed various compounds that actively exhibit ion channel structures and functions. As part of the design, chemists try to add functions such as pore size, ion or substitute selectivity, and voltage or ligand gating. In the early studies, researchers imitated natural compounds, such as valinomycin or gramicidin, for the structural framework of synthetic channels. Valinomycin is a macrocyclic polypeptide used in the transport of potassium, as an antimicrobial peptide. Gramicidin is also a polypeptide and forms a b-helix structure in the lipid monolayer; within the bilayer, two gramicidin molecules form an end-to-end dimer, which in turn forms a transmembrane structure in the bilayer. These macrocyclic or dimer structures have been modeled for the design of synthetic channels, and chemists synthesized the mimicking structure using synthetic peptides or macromolecules. More recently, other functional materials such as DNA origami and carbon nanotubes have been proposed as potential candidates for artificial ion channels.
3. Nanopore Technology Meets DNA Computing and is Applied to Practical Sensing
Nanopores—pore-forming transmembrane proteins—are another strong candidate for the sensors of molecular robots. Sensing with nanopores has emerged as a method for single molecule detection. This method works by applying an electric potential that causes single molecules to be passed through the nanopore. The change in the ionic current over time is recorded, potentially allowing the direct collection of information about individual molecules in terms of size and mobility. Although this method can detect single molecules, the selectivity relies on the size compatibility between the nanopore and target molecules. An a-hemolysin (aHL), pore-forming toxin from Staphylococcus aureus, is commonly used as the biological nanopore. This protein has a 1.4 nm diameter pore that allows single stranded DNA (ssDNA) to pass but blocks double-strand DNA (dsDNA), suggesting that the nanopore has precise size selectivity for DNA/RNA detection. For the detection of smaller molecules, a molecular adapter such as cyclodextrin, which reduces the pore size, has been used DNA aptamers have also been applied as the molecular tag for selective detection because aHL cannot detect larger sized molecules. Conversely, we have studied much larger nanopores from five different protein families for precise nanopore detection. Recently, apromising application of nanopore measurements for DNA sequencing has also resulted from ardent efforts, with a nanopore sequencer being commercialized in 2015.
These functions or technologies of such molecular machines and autonomous systems have matured sufficiently that they can operate on their own. Regarding the synthetic ion channels, early studies focused on constructing a transmembrane structure mimicking natural ion channels. Then, the transport properties such as ion selectivity or gating probability were investigated. Although in most cases the functions were carried out in a single operation, the natural ion channels work in signal cascades, and ion transport can induce a biological reaction. In the next step, the synthetic ion channel will be loaded onto the interface of the molecular robot. DNA's logic operations face a similar situation. Single operations such as AND, OR, and NAND can be combined to make sophisticated systems. Therefore, recent compelling work has shown that combining nanopore and DNA logic operations can result in a system that can be integrated into the body and interface with molecular robots.
Full study: source





